Why not scrub CO2 from the sky?

Everyone who has thought about industrially driven climate change has at some point, however briefly, wondered why we can't solve the problem by pulling the unwanted carbon dioxide back out of the air. Surely, if burning fossil fuels can blast so much extra carbon dioxide into the atmosphere, some other act of chemistry on an equally gargantuan scale ought to be able to recapture the gas.
Recent news out of the University of Southern California suggests that science is getting closer to making that antidote to global warming feasible. A close look at the technology, however, also shows why it would probably not be a perfect remedy all by itself.
Finding ways to draw carbon dioxide (CO2) out of the air is not in itself a terribly difficult problem. A variety of substances, such as calcium hydroxide, sodium hydroxide, and aminosilicas will under the right conditions spontaneously adsorb the gas. Submarines and spacecraft have long used filtration systems based on that principle to help keep their air supplies breathable.
But replicating that success on a massive industrial scale to offset the emissions from burning coal and oil gets problematic. Unless one has a bottomless, cheap supply of the sorbent material (and a place to store it once used), recycling it becomes a priority. Driving the CO2 back out of the sorbent usually entails raising its temperature, often by hundreds of degrees Celsius, which drives up the cost prohibitively.
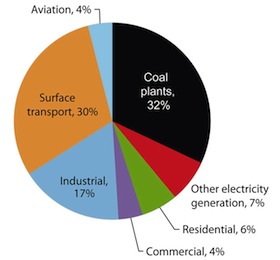
Because of pressures to curtail CO2 release, the energy industry has nonetheless been working on chemical systems based on ammonia or amines for scrubbing the carbon out of flue gases at coal- or oil-burning power plants. Currently, these approaches often cost about $100-150 per ton of CO2 abated, though expectations are that as the technology matures, costs will fall into the $35-50 per ton range.
Even so, that technology can only be fitted to stationary, concentrated sources of carbon emissions: adapting it for use on CO2 sources that are more mobile (like cars) or more diffuse (like cooking stoves and home heaters) isn't practical or affordable. It would therefore miss about half of all CO2 emissions from human activities. The ideal version of carbon capture would work independently of the sources. It would -- somehow affordably -- process the open air.
Air (re)conditioning
The latest advance in that direction has now come from Alain Goeppert, G. K. Surya Prakash, Nobel laureate George A. Olah and their colleagues at USC's Lokar Hydrocarbon Research Institute, as reported in the Nov. 21, 2011 issue of the Journal of the American Chemical Society. They have developed a new adsorbent plastic material that they maintain is inexpensive to produce and so voraciously hungry for CO2 that, until it fills up, it can thoroughly strip all the CO2 out of any surrounding air. Moreover, a temperature of only 85 °C is sufficient to release the CO2 and leave the plastic ready for reuse.
Their material consists of a highly branched organic compound called a polyethylenimine (PEI) that has been coated onto grains of silica, so that it offers lots of surface area. In humid, room-temperature air, each molecule of PEI reacts with CO2 and shifts into a stable bicarbonate form. (Even under more arid conditions, the PEI continued to take up CO2 well.) In tests, the material took up a bit less than 10 percent of its own weight in CO2 (1.71 mmol CO2 per gram of adsorbent).
The USC lab's technique is the latest of many developed to remove CO2 from air, and much more work still needs to determine whether it -- or something else yet to be invented -- is practical enough to deploy widely. Klaus S. Lackner of Columbia University has been a pioneer in the field, and Global Research Technologies, a company he founded, is attempting to commercialize his approach based on a carbonate polymer material. Other efforts (and compounds) are the focus of work at the Georgia Institute of Technology, Brookhaven National Laboratory, the University of Calgary, the Swiss Federal Institute of Technology, and elsewhere.
Assuming that any such approach pans out, what impact might it have on climate change? In a 2010 article on the subject for Scientific American, Lackner extrapolated from his technology to envision a time when 10 million air capture machines around the world, each extracting 10 tons of CO2 a day, would affordably collect 36 gigatons of CO2 per year. That would be enough to reduce the atmospheric concentration of CO2 by 5 parts per million annually -- more than twice the current rate at which CO2 is being added today. Lackner estimated that the initial costs might be about $200 per ton of CO2, but that as the technology matured, they might fall to around $30 per ton.
What technology alone can't do
Appealing as those projections sound -- and they do sound encouragingly good -- they shouldn't suggest that effective carbon capture makes global warming trivial to solve. First, developing and deploying the technology at that literally world-altering scale will take decades. Even a transient period of higher global temperatures could disrupt some ecosystems, contribute to natural disasters, and cause other headaches.
Then there's the issue of cost. An abatement cost of $35 per ton of CO2 may be reasonable; it should raise the cost of electricity by only a couple of cents per kilowatt-hour. But in recent U.S. policy discussions about cap-and-trade CO2 credits and carbon taxes, the idea of setting prices at even $20 per ton or less has met intense opposition. Who will be willing to foot the bill for building and operating the massive carbon capture infrastructure that Lackner described? As critics of carbon capture have argued for years, it might be less expensive simply to shift away from fossil fuels and to solar, wind, or other carbon-light energy sources.
Successful carbon capture also obliges us then to do something with the collected CO2. As Goeppert, Prakash, Olah, and Lackner have variously written, that CO2 has many industrial applications, including potential uses as an alternative to petrochemicals in the manufacture of polymers and fuels. A sizeable amount of it, however, will probably need to be sequestered -- for example, by pumping it underground into tapped-out oil fields or other sealed geological formations.
Yet carbon sequestration is still barely past its infancy. The Sleipner project has been storing more than 10 million metric tons of CO2 in formations under the North Sea off the coast of Norway for the past decade, and dozens of other projects are in various stages of execution or planning. But effective sequestration will need to work indefinitely, and concerns have arisen that at least some sites under consideration might not only leak CO2 but could lead to contamination of aquifers. Geologists are still determining how many formations around the world would be suited to sequestering carbon over the long term.
None of these objections means that scrubbing CO2 out of the atmosphere won't work well in practice. That the technology would allow the capture of CO2 to be delinked from the production of CO2 might actually be a point in its favor: it would pose less of an immediate thorn in the side to powerful energy interests and it might be more appealing this way to entrepreneurs. Removing CO2 from the air may in fact turn out to be an essential adjunct to curbs on CO2 production, such as switching to solar power: that combination might turn out to be the best hope for keeping greenhouse gas concentrations below catastrophic levels.
Even so, carbon capture doesn't represent a simple tech solution to climate change that can bypass the political and economic obstacles that have dogged the issue to date. The military theorist Carl von Clausewitz observed, "War is the continuation of politics by other means." The same can probably be said for technology.
•
Image: The earth's atmosphere, as photographed from the International Space Station. (Credit: ISS/NASA)
This post was originally published on Smartplanet.com
